High-Resolution AAV Profiling via Mass Photometry
Rapid Data Delivery
Get your full analytical report within
48 hours
<10 ul Sample
Cost-Effective
Performance Comparison: MP vs Traditional Methods
| Metric | Mass Photometry | AUC/TEM |
|---|---|---|
| Sample Volume | <10 ul | >500ul |
| Throughput | Rapid (Minutes) | Slow (Days) |
Population Distribution
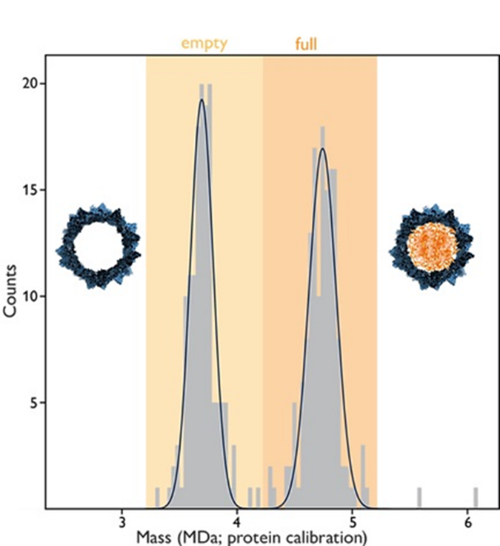
At INNOVAAVECTOR, we leverage cutting-edge Mass Photometry (MP) to provide rapid, precise, and cost-effective quantification of empty, full, and partially filled capsids. Get the data you need to advance your gene therapy program without the bottlenecks of traditional methods.
The MP Advantage:
Ultra-Low Sample Consumption: Achieve comprehensive results with as little as 10 µL of sample. Preserve your vector for what matters most.
Unrivaled Turnaround Time: From sample receipt to detailed reporting in under 48 hours. Real-time single-particle analysis in minutes.
Cost-Efficiency: A high-resolution alternative to Analytical Ultracentrifugation (AUC) and Cryo-EM at a fraction of the cost.
High-Resolution Profiling: Distinctly quantify Empty, Partial (truncated/sub-genomic), and Full populations with unmatched sensitivity.
Precision at the Single-Molecule Level
Our Mass Photometry platform operates by measuring the light scattering of individual AAV particles. This allows for the direct determination of molecular mass in a native buffer environment. The result? A clear, histogram-based distribution of your capsid loading, ensuring the purity and potency of your final product.
GMP-Ready Analytics
Ensure the highest standards for your clinical programs. Our Mass Photometry service is also available fully GMP compliant, making it an indispensable tool for quality control, batch release testing, and in-process monitoring of AAV manufacturing. We provide the regulatory-grade data integrity required to move your therapy from the bench to the patient.
