Flexible, fully customized cell banking solutions to support the development and manufacturing of biologics and advanced therapies
What we offer
- GMP production of Master Cell Banks (MCB) as the primary, fully characterized source cell stock for all future manufacturing runs.
- Generation of Working Cell Banks (WCB) derived from the MCB to ensure a consistent and reproducible cell supply over time.
- A two‑tier MCB/WCB system that secures a reliable cellular substrate across the entire product life cycle.
Why it matters
- GMP cell banks ensure genetic stability, traceability and help reduce the risk of drift, contamination and out‑of‑spec production lots.
- A robust MCB/WCB strategy underpins clinical and commercial manufacturing while meeting FDA, EMA and other regulatory expectations.
Our added value
- Modular processes: we tailor batch size, vial configuration, cell concentration, QC testing panel and documentation package to your specific stage and application (discovery, clinical, commercial).
- Co‑designed cell banking plan: we work with you to define characterization assays, release criteria and long‑term banking strategy for MCB and WCB.
- Integrated services: optional process development, scale‑up, long‑term cryostorage and regulatory support bundled in a single service offering.
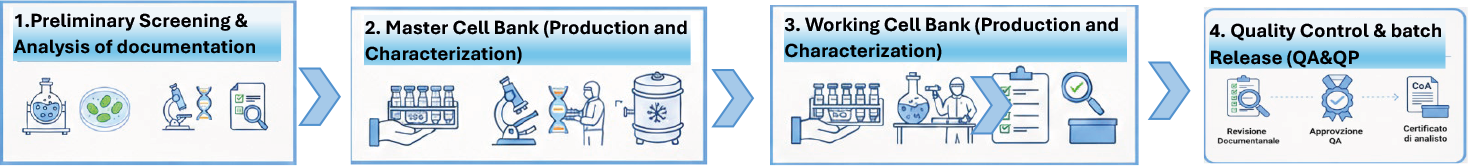
How we work (short flow)
You can visualize this as a 3–4 step graphic on a slide.
- Initial assessment
- Review of your cell line, target indication and intended use (research, clinical, commercial).
- Master Cell Bank design
- Definition of GMP protocol, culture conditions, number of vials and QC/characterization strategy.
- GMP manufacturing of MCB and WCB
- Execution in GMP cleanrooms with testing for identity, purity, sterility, genetic stability and viability.
- Release, storage and ongoing support
- Complete batch documentation, certificates of analysis, secure long‑term storage and support for regulatory submissions
